Jump to paragraph
Introduction
Neurodegenerative disorders are characterized by the progressive and irreversible loss of neurons from specific regions of the brain, spinal cord, or both Prototypical neurodegenerative disorders, including Parkinson disease (PD) and Huntington’s disease (HD), where the loss of neurons from structures of the basal ganglia results in abnormalities in the control of movement; Alzheimer’s disease (AD), where the loss of hippocampal and cortical neurons leads to impairment of memory and cognitive ability.
These disorders are relatively common and represent a significant medical and societal problem. They are primarily disorders of later life, developing in neurologically normal individuals, although childhood-onset forms of disorders are recognized.
Currently, available therapies for neurodegenerative disorders alleviate the disease symptoms but do not alter the underlying neurodegenerative process. The available symptomatic treatments for AD, HD, and ALS are much more limited effectiveness. On the horizon are pharmacological treatments aimed at preventing or retarding the progression of neuron-degeneration.
Alzheimer’s Disease
Alzheimer’s disease is the most common cause of dementia in the elderly, with cerebrovascular disease. Most cases of Alzheimer’s occur after the age of 50, with a progressive increase in incidence with increasing age. Most cases are sporadic, but in roughly 10% of patients, there is a family history of dementia. Alzheimer’s disease embraces the two conditions previously designated senile dementia (dementia occurring after 65 age) and presenile dementia (dementia has an onset before 65 years of age).

The brain region most vulnerable to neuronal dysfunction and cell loss in AD is the medial temporal lobe, including the entorhinal cortex and hippocampus. Typical early AD symptoms are due to dysfunction of these structures resulting in anterograde episodic memory loss: repeated questions, misplaced items, missed appointments and forgotten details of daily life.
A typical patient presents with memory dysfunction that is noticeable by the patient or family members, but not severe enough to impair daily function.
Current diagnostic criteria for AD requires the presence of dementia (i.e., cognitive impairments sufficient to reduce function).
The later stages of the disease are characterized by increasing dependence and progression toward the akinetic-mute state that typifies end-stage neurologic disease. Death, most often from a complication of immobility such as pneumonia or pulmonary embolism, usually ensues within 6-12 years of onset.
At present, the diagnosis of Alzheimer’s disease is based on the clinical assessment of the patient. Structural neuroimaging and appropriate laboratory tests are used to exclude other disorders that may mimic AD.
Laboratory measures to specifically identify AD, including analysis of biomarkers such as CSF or serum factors, genetic testing, and molecular or functional neuroimaging, are likely to be incorporated into Alzheimer’s disease. Diagnostic criteria may enhance the sensitivity of diagnosis, especially at the early stages of the disease.
Pathophysiology of Alzheimer’s Disease

Alzheimer’s disease attacks nerves and brain cells as well as neurotransmitters. The brain in AD may be grossly normal in the earlier stages of the disease. The atrophy is most evident in the frontal, temporal, or parietal lobes but usually involves all cortical areas to some degree. Microscopic changes include neurofibrillary tangles, which appear as coarse, filamentous aggregates within the cytoplasm of neurons. They are found in the neocortex, hippocampus, basal forebrain, and brain stem.
- FORMATION OF NUMEROUS NEUROFIBRILLARY TANGLES
The neurofibrillary tangles are composed of insoluble, protein-rich paired helical filaments. These protein-rich paired helical filaments occur to form characteristic senile plaques, which appear as aggregates of coarse, tortuous neurites in the neuropil of the cerebral cortex.
The senile plaques contain a central amyloid core composed of an amyloid core made of beta-amyloid.
The simple presence of plaques or tangles is not, by itself, specific for AD, because such structures are also frequently found in the brains of normal elderly individuals. Instead, it is the number of plaques and tangles in neocortical areas in the setting of dementia that allow one to make a diagnosis of Alzheimer’s disease.
- ACCUMULATION OF SENILE PLAQUES (Β-AMYLOID ACCUMULATIONS)
The deposition of amyloid, derived from the breakdown of a protein known as an amyloid precursor protein(APP), is a consistent feature of AD. The breakdown product, known as β-amyloid, is a prominent component of both neurofibrillary triangles and senile plaques found in the brains of AD patients, and usually present within the walls of cerebral blood vessels. β-amyloid has also been shown to be toxic to neurons in cell cultures. There is evidence that amyloid deposition plays a primary role in the development of AD.
The expression of specific alleles of apoprotein (apoE) has been demonstrated in AD. It has been suggested that apoE may be involved in the transport or processing of the β-amyloid precursor protein. ApoE ε4 is reported to bind better to β-amyloid than other forms of apoE and may contribute to enhanced amyloid fibril formation.
- LOSS OF CORTICAL NEURONS, PARTICULARLY CHOLINERGIC NEURONS
The most striking neurochemical disturbance in AD is a deficiency of acetylcholine. The anatomical basis of the cholinergic deficit is atrophy and degeneration of subcortical cholinergic neurons, particularly those in the basal forebrain that provide cholinergic innervation to the cerebral cortex.
The selective deficiency of ACh in AD, as well as the observation that central cholinergic antagonists such as atropine can induce a confusional state that bears some resemblance to the dementia of AD, is critical in the genesis of AD.
Although viewing AD as a “cholinergic deficiency syndrome” is akin to the “dopaminergic deficiency syndrome,” it is essential to note that the deficit in AD is far more complex. Alzheimer’s disease involves multiple neurotransmitter systems, including glutamate, 5-HT, and neuropeptides, and there is the destruction of not only cholinergic neurons but also the cortical and hippocampal targets that receive cholinergic input.
Environmental Factors Affecting Alzheimer’s Disease
- Smoking
- Certain Infection
- Toxins
- Cholesterol-Lowering Drugs

Risk Factors Of Alzheimer’s Disease
Morphologic changes identical to those seen in AD are almost invariably present in patients with Down syndrome who survive beyond 40. Despite extensive research in recent years, the cause of AD is unknown, although many factors associated with the development of the disease have been identified:
- Increasing Age
- Down Syndrome
- Head Injury
- Obesity
- High Blood Pressure
Genetic Factors
Genetic factors may play a role in developing some cases of Alzheimer’s, as evidence by the occurrence of familial cases. Many investigations have implicated genetic abnormalities on different chromosomes of both sporadic and familial AD. Most familial cases are linked to a mutation on chromosomes 14. The function of this gene product is not known. The genetic heterogeneity noted thus far suggests that no single genetic defect is likely to be responsible for all cases of AD.
Signs and Symptoms
- Loss of memory: forgetting things often or struggling to retain new information affect day to day abilities.
- Difficulty performing familiar tasks: forgetting basic tasks, how to do something that has been done their whole life.
- Changes in mood and behavior: exhibiting severe mood swings.
- Problem speaking: forgetting words or using words that don’t fit the content.
- Disorientation in space and time: getting lost in a familiar place or not knowing the date or day of the week.
- Impaired judgment: not recognizing a problem that needs attention.
- Misplacing things: putting things in wrong or strange places.
- Change in personality: behavior that is suspicious, fearful, confused, or irritated.
- Loss of initiative: losing interest in family, friends, and activities.
Treatment of Alzheimer’s Disease
At present, none of the currently available therapeutic agents have been shown to alter the underlying neurodegenerative process, and no disease-modifying therapy for AD is accessible. Aggressive attempts to develop drugs targeting Aβ, apoE and other molecules involved in AD pathogenesis are underway. However, current treatment is aimed at merely alleviating symptoms.
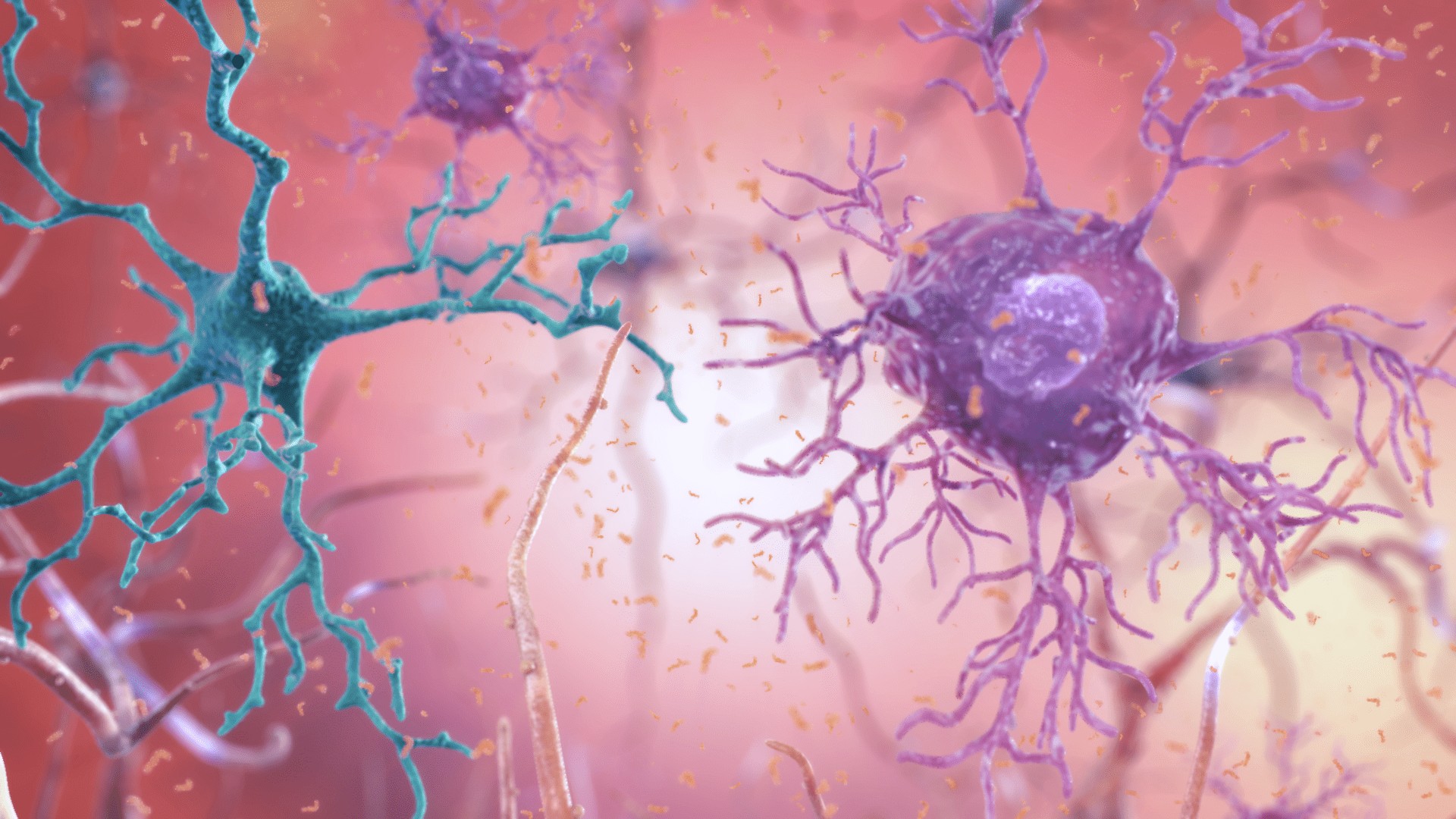
Dementia of the Alzheimer Type
- Accumulation of senile plaques (β-amyloid accumulations)
- Formation of numerous neurofibrillary tangles
- Loss of cortical neurons, particularly cholinergic neurons
Current therapies are aimed at either improving cholinergic transmission within the CNS or preventing excitotoxic actions resulting from overstimulation of NMDA-glutamate receptors in selected brain areas.
Acetylcholinesterase Inhibitors
Numerous studies have linked the progressive loss of cholinergic neurons and, evidently, cholinergic transmission within the cortex to the memory loss that is a hallmark symptom of AD. It is postulated that inhibition of acetylcholinesterase (AChE) within the CNS will improve cholinergic transmission, at least at those neurons that are still functioning.
Currently, four reversible AChE inhibitors are approved for the treatment of mild to moderate AD. They are:
- Donepezil
- Galantamine
- Rivastigmine
- Tacrine
Common adverse effects include nausea, diarrhea, vomiting, anorexia, tremors, bradycardia, and muscle cramps, all of which are predicted by the actions of the drugs to enhance cholinergic neurotransmission. Unlike the others, tacrine is associated with hepatotoxicity.
NMDA-receptor Antagonist
- Antagonists of the NMDA-glutamate receptor are frequently neuroprotective, forestalling the loss of neurons following ischemic and different wounds.
- Memantine is a dimethyl adamantane subordinate. Memantine acts by physically blocking the NMDA receptor–associated ion channel, but, at therapeutic doses, only a fraction of these channels are blocked. Memantine has been shown to slow memory loss in both vascular-associated and Alzheimer’s dementia in patients with moderate to severe cognitive losses.
- However, there is no evidence that memantine prevents or slows the neurodegeneration in patients with AD or is more effective than the AChE inhibitors.
- Memantine is well tolerated, with few dose-dependent adverse events.
- Expected side effects, such as confusion, agitation, and restlessness, are indistinguishable from the symptoms of AD.
- Memantine is often given in combination with an AChE inhibitor.
Conclusion
Clinical features of AD include a progressive impairment of memory and other cognitive functions. Symptoms may be subtle at first and quickly confused with depression, another clinically important disease in the elderly. Cognitive impairment continues inexorably, usually over 5 to 15 years, resulting in complete disorientation and loss of language and other higher cortical functions. Death usually results from intercurrent bronchi pneumonia or other infections.
The human and societal cost of Alzheimer’s disease is staggering. However, there is hope that earlier and better diagnosis, increased knowledge of its explanation (with support of the patient and family throughout the stages of the disease), effective symptomatic drugs, and potentially effective disease modification strategies will have a dramatic impact on the number of persons affected in the future, and the quality of life of persons currently affected. The fast pace of research and development in AD is unique in neurological history and should lead to a better future for aging populations.
Bonus video: Inside Alzheimer’s disease
References
Roger Walker Clinical Pharmacology